Introduction
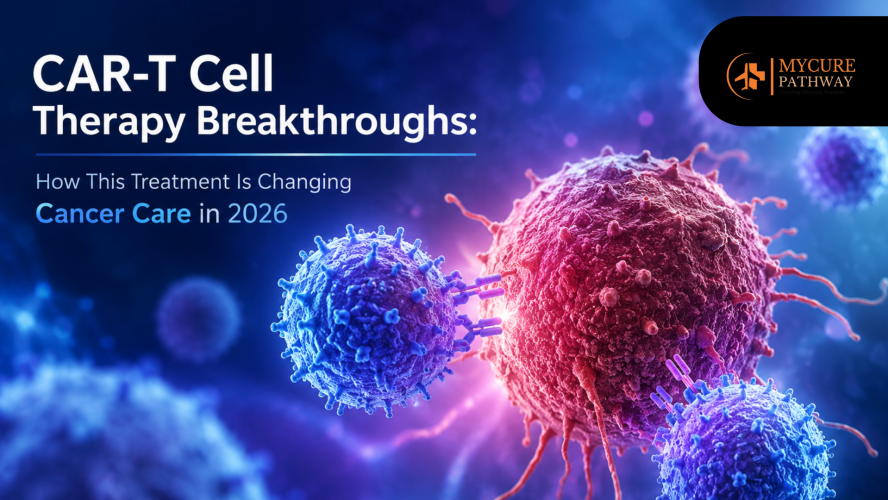
Cancer treatment has entered a new era in 2026, and one of the biggest medical innovations driving this transformation is CAR-T Cell Therapy. Once considered an experimental option for patients with limited treatment choices, CAR-T has now become one of the most promising forms of advanced cancer treatment in 2026 for blood cancers such as leukemia, lymphoma, and multiple myeloma.
Unlike traditional chemotherapy or radiation, CAR-T Cell Therapy uses the patient’s own immune system to recognize and destroy cancer cells. This personalized immunotherapy has delivered remarkable outcomes, especially for patients with relapsed or treatment-resistant cancers. Recent CAR-T cell therapy breakthroughs are also improving treatment speed, reducing side effects, and expanding eligibility for more patients worldwide.
In this guide, we explore how CAR-T Cell Therapy is revolutionizing cancer care, who can benefit from it, treatment costs, and why many international patients are now considering CAR-T therapy in India.
What Is CAR-T Cell Therapy?
CAR-T Cell Therapy stands for Chimeric Antigen Receptor T-cell Therapy. It is a highly personalized form of immunotherapy where doctors genetically modify a patient’s T-cells to identify better and attack cancer cells.
How CAR-T Cell Therapy Works
1. T-Cell Collection
Doctors collect T-cells from the patient’s blood through a process called leukapheresis.
2. Genetic Engineering
These T-cells are modified in a laboratory to express special receptors called CARs (Chimeric Antigen Receptors).
3. Cell Expansion
The modified cells are multiplied by millions.
4. Reinfusion
The engineered CAR-T cells are infused back into the patient’s bloodstream.
5. Cancer Cell Destruction
The CAR-T cells locate and kill cancer cells more effectively than normal immune cells.
This process makes CAR-T Cell Therapy a “living drug” because the modified cells can continue working inside the body for months or even years.
Why CAR-T Cell Therapy Is a Major Cancer Breakthrough in 2026
Several CAR-T cell therapy breakthroughs are changing cancer care in 2026.
1. Faster Manufacturing Time
Previously, manufacturing CAR-T cells could take 3–6 weeks, delaying treatment for aggressive cancers. In 2026, newer production systems and automated labs will significantly reduce wait times.
This is especially important for patients with rapidly progressing leukemia or lymphoma.
2. Better Safety Profiles
Early CAR-T treatments carried risks such as:
- Cytokine Release Syndrome (CRS)
- Neurotoxicity
- Fever and immune complications
New-generation CAR-T therapies now include:
- Improved monitoring protocols
- Safer cell engineering
- Better toxicity management
These improvements are making CAR-T Cell Therapy safer and more accessible.
3. Expansion Beyond Blood Cancers
Initially, CAR-T Cell Therapy was mainly approved for blood cancers. In 2026, research is expanding into:
- Brain tumors
- Lung cancer
- Ovarian cancer
- Pancreatic cancer
- Colorectal cancer
While blood cancers still show the best results, the future of CAR-T is expanding rapidly.
4. Off-the-Shelf CAR-T Products
Traditional CAR-T is personalized, requiring cells from each patient.
A major CAR-T cell therapy breakthrough in 2026 is the development of allogeneic or off-the-shelf CAR-T therapy, where donor cells are used.
Benefits include:
- Faster treatment access
- Lower manufacturing complexity
- Potential cost reduction
This could make CAR-T Cell Therapy more affordable globally.
Which Cancers Can Be Treated with CAR-T Cell Therapy?
Currently, CAR-T Cell Therapy is most successful in hematologic cancers.
Leukemia CAR-T Treatment
Leukemia CAR-T treatment has shown impressive remission rates, particularly for:
- Acute Lymphoblastic Leukemia (ALL)
- B-cell Acute Lymphoblastic Leukemia
- Chronic Lymphocytic Leukemia (CLL)
Patients with relapsed or refractory leukemia are often strong candidates.
Immunotherapy for Lymphoma
Immunotherapy for lymphoma using CAR-T has changed treatment outcomes for:
- Diffuse Large B-Cell Lymphoma (DLBCL)
- Follicular Lymphoma
- Mantle Cell Lymphoma
- Primary Mediastinal B-cell Lymphoma
Many patients who failed chemotherapy have achieved durable remission with CAR-T.
Multiple Myeloma
CAR-T is also increasingly used for:
- Relapsed multiple myeloma
- Treatment-resistant myeloma
New targets like BCMA are improving patient outcomes.
Who Is Eligible for CAR-T Cell Therapy?
Not every patient qualifies for CAR-T Cell Therapy.
Eligibility often depends on:
- Cancer type
- Stage of disease
- Previous treatment history
- Organ function
- Performance status
Common candidates include:
- Relapsed cancer patients
- Refractory cancer patients
- Patients who failed chemotherapy or stem cell transplant
A specialist evaluation is essential before proceeding.
CAR-T Therapy Cost: What Patients Should Expect
One of the biggest concerns for patients is the cost of CAR-T therapy.
Pricing varies significantly by country.
Approximate CAR-T Therapy Cost by Region
| Country | Estimated Cost |
| United States | $400,000–$500,000+ |
| UK/Europe | High, depending on access |
| India | More cost-effective options |
The total CAR-T therapy cost includes:
- Cell collection
- Lab modification
- Hospitalization
- ICU monitoring
- Medications
- Follow-up care
Because of these high costs, many international patients are exploring affordable options abroad.
Why Patients Are Choosing CAR-T Therapy in India
CAR-T therapy in India is gaining global attention due to affordability, medical expertise, and growing infrastructure.
Benefits include:
1. Lower Treatment Cost
India offers comparatively lower treatment expenses than Western countries.
2. Experienced Cancer Specialists
India has internationally trained hematologists, oncologists, and transplant specialists.
3. Advanced Hospitals
Many hospitals now offer advanced cell therapy units and precision oncology care.
4. Medical Travel Support
International patients can receive end-to-end assistance.
Platforms like MyCure Pathway help global patients explore treatment options, hospital coordination, doctor consultations, visa support, and treatment planning for complex procedures such as CAR-T Cell Therapy.
Challenges of CAR-T Cell Therapy
Despite its success, CAR-T Cell Therapy still faces limitations.
High Cost
Manufacturing remains expensive.
Limited Access
Not all hospitals offer CAR-T.
Side Effects
Potential complications include:
- CRS
- Neurological toxicity
- Infection risk
Solid Tumor Challenges
Success in solid tumors is still under active research.
However, ongoing CAR-T cell therapy breakthroughs are addressing these challenges rapidly.
Future of CAR-T Cell Therapy Beyond 2026
The future of CAR-T Cell Therapy looks highly promising.
Emerging innovations include:
Dual-target CAR-T
Targets multiple cancer antigens.
Armored CAR-T Cells
Designed to survive hostile tumor environments.
In Vivo CAR-T
Cells engineered directly inside the patient’s body.
AI-Driven Cell Design
Artificial intelligence is helping optimize cell engineering and patient matching.
These innovations may further transform advanced cancer treatment in 2026 and beyond.
Frequently Asked Questions (FAQs)
Is CAR-T Cell Therapy a cure for cancer?
Not always. However, CAR-T Cell Therapy can lead to long-term remission in some patients, especially with blood cancers.
How long does CAR-T treatment take?
Typically:
- Cell collection: 1 day
- Manufacturing: 2–4 weeks
- Hospital stay: 1–3 weeks
Is CAR-T therapy painful?
The infusion itself is similar to a blood transfusion, but side effects may require close monitoring.
What is the success rate of leukemia CAR-T treatment?
Success varies, but remission rates can be very high in eligible leukemia patients.
Is CAR-T therapy available in India?
Yes, CAR-T therapy in India is increasingly available at specialized cancer centers.
Patients often seek guidance through medical facilitators such as MyCure Pathway to identify hospitals and streamline treatment access.
Conclusion
The evolution of CAR-T Cell Therapy is one of the most exciting developments in modern oncology. With major CAR-T cell therapy breakthroughs, safer protocols, broader cancer applications, and growing accessibility, this treatment is redefining what is possible in cancer care.
For patients facing difficult diagnoses such as leukemia or lymphoma, CAR-T Cell Therapy offers renewed hope where conventional treatments may no longer work.
As research continues, advanced cancer treatment 2026 is no longer just about managing disease—it is increasingly about personalized precision medicine that gives patients a second chance at life.
For international patients exploring CAR-T therapy in India, early consultation with experienced oncology teams and trusted medical facilitators can make the treatment journey smoother, more informed, and more accessible.